MEMS AS®
Medication Adherence Software
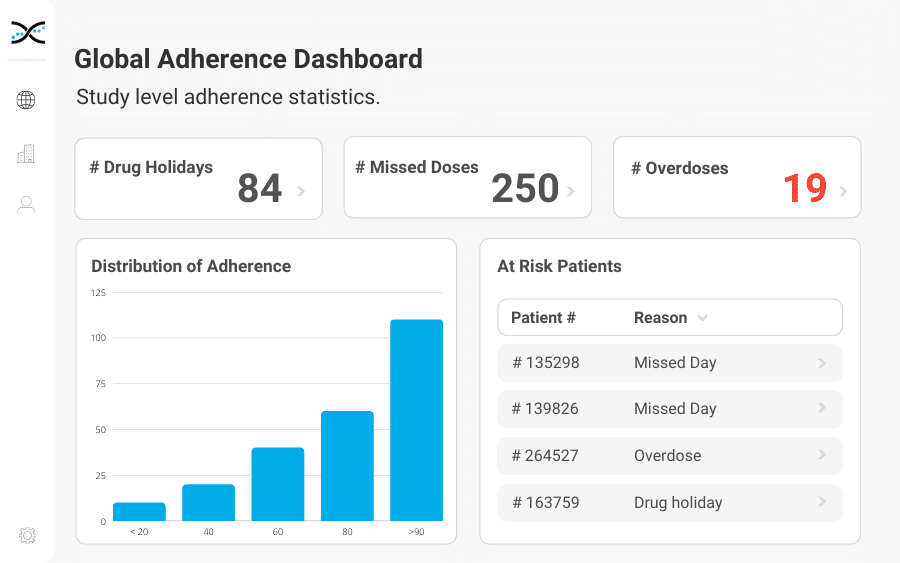
This powerful medication adherence software is about much more than understanding whether participants are taking their medications on time. With its real-time, user-friendly adherence dashboards, It's propelling drug developers to new heights as they strive to achieve the best possible outcomes for their clinical trial participants.


MEMS AS
Measure Adherence Precisely with Medication
Adherence Software.
When conducting clinical trials for new medications, ensuring adherence to medication regimens is vital to the study's success. The effectiveness of a medication can be significantly impacted by non-adherence, making it difficult for researchers to measure the true efficacy of the treatment under study. However, measuring and promoting desired patient behavior can be challenging for study investigators. It's not always easy to keep track of every participant's medication schedule and ensuring that patients follow their prescribed regimens can be challenging. That's where medication adherence software comes in.
Our innovative medication adherence software, MEMS AS, is powered by proprietary algorithms and digitally enabled medication adherence packaging and devices. This allows study investigators to measure and monitor their participants' medication-taking habits accurately. With its powerful analytics, the MEMS AS provides an intuitive user interface and elegant reporting to present a complete visual picture of patient medicine-taking behavior.
Trusted by the world's leading pharmaceutical firms, MEMS AS medication adherence software has become the gold standard for measuring medication adherence in clinical trials. Its simple, powerful, and intuitive design helps study investigators stay on top of their game and achieve the best possible results for participants. With MEMS AS, researchers can rest assured that they are getting the most accurate and up-to-date information on their trial participants' medication behaviors. This data helps investigators make well-informed decisions that ensure optimal study performance, reduce costs, and increase positive regulatory outcomes.

FEATURES
Cloud-Based Medication Adherence Software.
Our secure, cloud-based medication adherence software, MEMS AS, is perfect for researchers that require real-time medication adherence insights. Driven by over seventy proprietary algorithms, MEMS AS interrogates the medication event data from digitally enabled medication adherence packaging and devices to provide powerful dashboard visualizations highlighting missed days and drug holidays. With MEMS AS in their toolkit, researchers can rest assured that they have access to precise and accurate data to underpin safety and efficacy calculations.
- Fully Scalable
- ISO27001 Certified Datacenter
- HIPAA & GDPR Compliant
- IRT, EDC & DCT Integration

FEATURES
Unparalleled Accuracy and Reliability.
Boasting an unmatched accuracy rate of 100%, our medication adherence software sets the standard for medication adherence software. MEMS AS comes equipped with ready-made dashboards that provide critical insights into adherence at the participant, study, and site levels, giving researchers a frictionless solution for monitoring medication adherence. By revolutionizing medication adherence monitoring, MEMS AS is changing the game for researchers, allowing them to offer personalized medication management and coaching for participants. With MEMS AS, researchers can ensure the best possible outcomes.
- Mature Solution
- Used in 200+ Drug Trials
- Over 1Mil participants monitored
- Cited in 800+ Papers

FEATURES
Suitable for Oral Dose, Injectable, Inhalable Drugs & More.
We offer a comprehensive approach to medication adherence monitoring that includes medication adherence packaging and devices from a broad range of industry-leading vendors. This enables us to provide solutions for all routes of administration, including oral dose drugs, injectables, inhalable, and more. Our approach ensures that medication adherence can be monitored across all types of medication and delivery methods, offering unparalleled accuracy and convenience for researchers.
- Participant Acceptability High
- User-friendly

FEATURES
Improve Adherence to Prescribed Medication.
Enabling optimal adherence in clinical trials is crucial for obtaining optimal participant outcomes and maintaining the integrity of research. Our adherence packaging and devices are designed to be user-friendly and do not add any burden to participants, as they only need to take their medications as prescribed. Additionally, participants can leverage the benefits of MEMS® Mobile, which allows them to schedule medication reminders and remain on track with their medication.
- User-Friendly App
- IoS & Android Compatible
- Available in 25 Languages
- 20K+ Users in 30 Countries

FEATURES
Approach Backed by Regulators.
The "Enrichment Strategies for Clinical Trials to Support Determination of Effectiveness of Human Drugs and Biological Products" guidance from the FDA emphasizes the importance of medication adherence and calls on the research industry to prioritize it. Specifically, the FDA recommends using medication adherence packaging to promote participants' compliance with their prescribed medication regimen
- ICH GCP Compliant
- FDA 21 CFR part 11 Compliant

OUR CLIENTS
The Go-To Solution for Pharma Companies
Some of the world's leading pharmaceutical companies have embraced our medication adherence software. From global giants to niche players, these organizations have recognized the value of our innovative solutions for enhancing medication adherence, reducing costs, and improving patient outcomes. It's an honor to partner with these remarkable brands, and we're proud to contribute to their efforts in advancing healthcare.

Medication Adherence Software →
Learn about our industry-leading adherence software for trials.

Medication Adherence Packaging →
Discover our range of medication adherence packaging.

Discover our range of medication adherence devices.
Got Questions?
Connect with an adherence expert.
Frequently Asked Questions
Medication adherence is a vital yet often overlooked aspect of successful research. That's why we've gone the extra mile to gather and organize the most frequently asked questions about this critical topic. Our goal is to empower researchers and patients alike with the knowledge they need to ensure medication adherence is never a hurdle to progress. So, without further ado, here are the answers you've been looking for!
The most accurate method of measuring medication adherence depends on several factors, including the medication type, the study design, and available resources. No single method is perfect, and each method has its own strengths and limitations. However, electronic monitoring using Medication Event Monitoring System (MEMS) devices combined with centralized, remote adherence monitoring software is considered the most accurate method for measuring medication adherence in clinical trials – and it is proven to be 97% accurate.
How electronic monitoring works
Trial participants are provided with medication adherence packaging or devices that include hidden trackers. For example, smart blister packs may have hidden microprocessors hidden in each cavity that activate when a pill is removed. Smart pill bottles, which also tend to have covert sensors in the cap, activate when the cap is removed from the bottle, conveniently registering the date, time, and in some cases, the location when the bottle has been opened.
Medication event data from the adherence packaging or device is synchronized with software that processes the data to highlight and present problematic patterns in dashboards for researchers. This approach takes the guesswork out of decoding adherence behaviors and helps researchers get to the participants that require coaching and support much sooner than counterpart methods.
Choosing the most accurate method for measuring medication adherence requires careful consideration of the study’s specific requirements, resources, and patient population. The selected method should provide reliable and valid data while minimizing patient burden and meeting ethical considerations.
In some studies, mobile apps designed to support medication adherence have shown promise in improving adherence and health outcomes. While the evidence is still emerging, several systematic reviews and meta-analyses have evaluated the effectiveness of mobile apps in promoting medication adherence.
A systematic review of 17 randomized controlled trials (RCTs) published in 2019 found that mobile apps were associated with improved medication adherence compared to usual care or no intervention [1]. The review also found that apps that provided feedback, reminders, and educational content were more effective than those that did not.
Another systematic review and meta-analysis of 29 RCTs published in 2020 found that mobile apps significantly improved medication adherence, self-efficacy, and health outcomes compared to usual care or no intervention [2]. The study also found that apps that included medication reminders, educational content, and self-monitoring features were more effective than those that did not.
References:
[1] De Vera, M. A., Ratzlaff, C., Doerfling, P., Kopec, J. A., & Gromala, D. (2019). Mobile app–based interventions to support self-management in individuals with rheumatoid arthritis: A systematic review and meta-analysis. Arthritis Care & Research, 71(10), 1292-1305.
[2] Liu, X., Guo, X., Li, Y., Jiang, J., Chen, X., Li, Z., … & Zhang, X. (2020). Mobile application interventions for medication adherence in patients with hypertension: A systematic review and meta-analysis. Journal of Cardiovascular Nursing, 35(4), 369-378.
There are several strategies that researchers can use to improve medication adherence in clinical trials. Here are three examples:
Patient education: Providing patients with clear and concise information about their medication, including its purpose, dosage, side effects, and potential interactions, can help improve medication adherence. Patients who understand the benefits and risks of their medication are more likely to take it as prescribed. Education can be delivered through various methods, such as printed materials, videos, or in-person counseling sessions.
Reminder systems: Using reminder systems, such as text messages, phone calls, email notifications, or calendarized reminders, like those featured in MEMS Mobile, can help improve medication adherence by prompting patients to take their medication at the right time. These systems can be customized to the patient’s specific needs and preferences and can be tailored to match the dosing schedule and medication regimen.
Support services: Providing patients with additional support services, such as medication counseling, peer support groups, or transportation assistance, can help address the barriers that prevent them from adhering to their medication regimen. These services can be delivered by healthcare providers or community-based organizations and can be tailored to the patient’s specific needs and preferences.
Non-adherence to medication in clinical trials can significantly negatively impact the study results, the interpretation of the findings, and, ultimately, patient health outcomes. Here are some examples of the potential impact of non-adherence in trials:
Reduced statistical power: this can reduce the statistical power of a clinical trial, making it more difficult to detect significant differences between the treatment and control groups. This can lead to inconclusive results, false-negative or false-positive findings, and wasting resources and time.
Bias in treatment effect estimates: can bias the treatment effect estimates, making them either over- or underestimated. This can lead to inaccurate conclusions about the efficacy or safety of the medication being studied, which can have profound implications for patient care.
Increased risk of adverse events: this can increase the risk of adverse events and medication-related complications, as patients who do not take their medication as prescribed may not achieve the desired therapeutic effect or may experience unintended side effects.
Decreased generalizability: this can decrease the generalizability of the study results, as patients who do not adhere to the medication regimen may differ from those who do in terms of their demographic characteristics, disease severity, or other factors that affect treatment response.
Increased costs: this can increase the costs of the study, as additional resources may be needed to identify and address non-adherence, such as more frequent monitoring, additional visits, or more intensive support services.
Overall, non-adherence is a significant challenge in clinical trials that can undermine the validity and reliability of the study results and compromise patient health outcomes. To minimize the impact of non-adherence, researchers should implement effective strategies to improve medication adherence and monitor adherence throughout the study period.
WEBINAR WITH MERCK & BIOGEN
Mitigating the Risk of Poor Adherence in Trials
Watch this live recording with adherence experts from Merck & Biogen to learn about their approach to mitigating the risk of poor adherence in trials.


Collaborating for Safer, More Efficient Trials.
By combining technology and partnerships, we are revolutionizing how medication adherence is monitored in clinical trials. Our unique adherence ecosystem brings together leading medication adherence packaging and devices and DCT, IRT, and EDC vendors, CROs, and CMOs to drive innovation.
