MEMS® Button Medication Event
Monitoring System
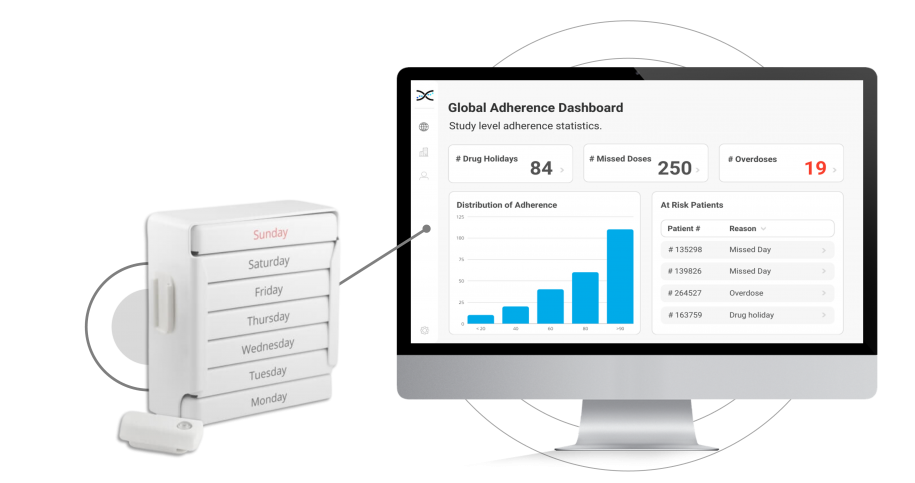
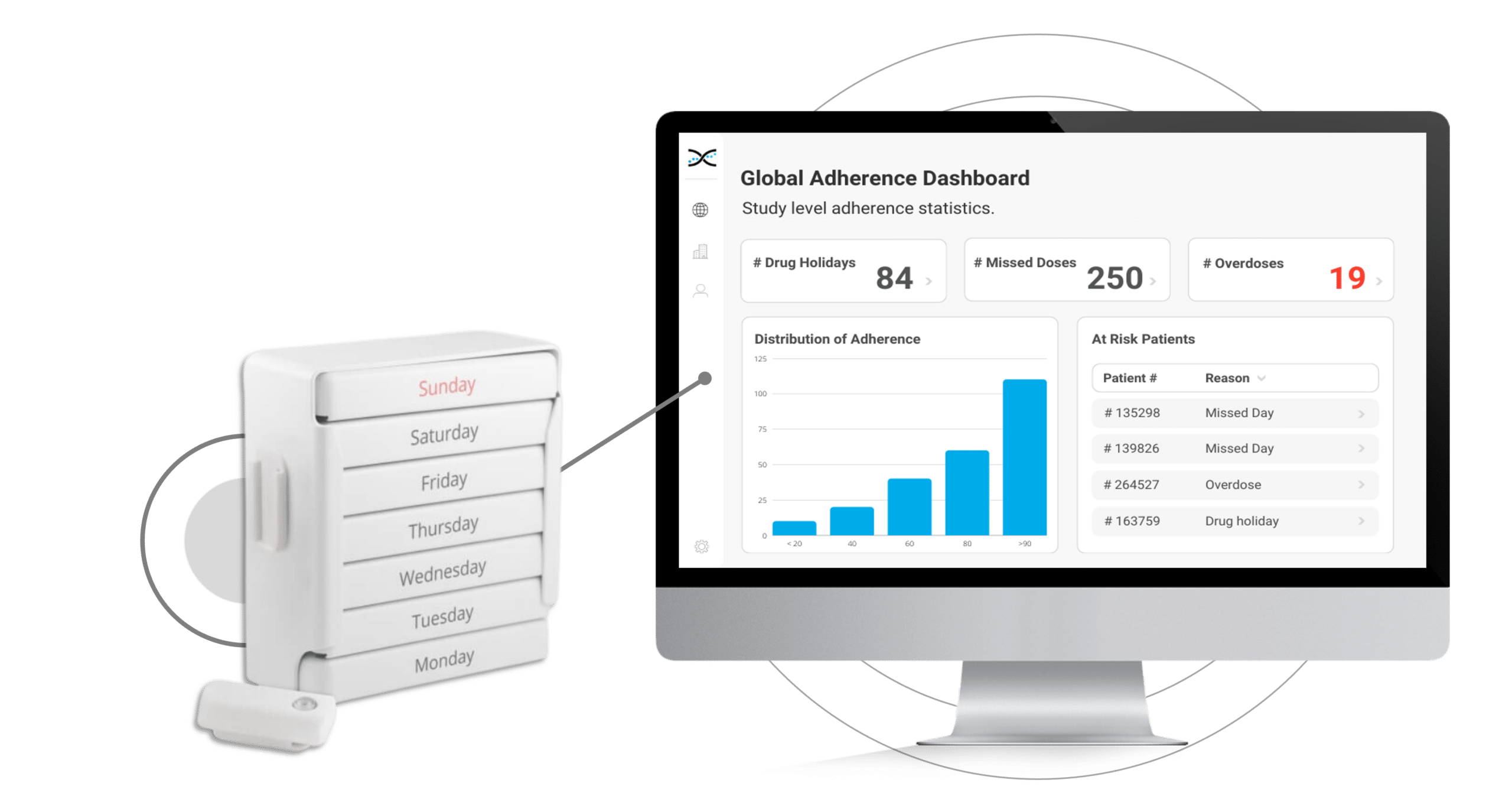
The MEMS Button is an electronic Medication Event Monitoring System used to monitor adherence in clinical trials. It works by incorporating a micro-electronic circuit that records the date and time each time the participant presses the button. It provides a straightforward and reliable method for monitoring adherence behaviors. This practical approach makes understanding how participants take their medications easier and provides access to user-friendly dashboards for better tracking and analysis of adherence data.

MEMS BUTTON
Improved Adherence is Just a Click Away
In addition to its compact design and compatibility with various medication containers, the MEMS Button offers a range of advanced features that make it a valuable tool for clinical trial research. For example, the MEMS Button has a programmable timer that can be used to record specific dosing intervals. This feature can be handy for medications that need to be taken at particular times of the day or for drugs with complex dosing regimens.
Furthermore, the MEMS Button's integration with AARDEX Group's medication adherence software, MEMS AS®, provides clinical researchers valuable insights into patient dosing patterns. The software includes a range of pre-built dashboards and ranked lists that allow researchers to track participant, site, and study adherence patterns easily. With these valuable insights at their fingertips, researchers can proactively identify adherence issues and take appropriate measures to ensure participants stay on track with their medication regimens.
The MEMS Button is a reliable and effective tool for promoting medication adherence in clinical trials. The MEMS Button enables researchers to produce more reliable and accurate study results by providing objective and reliable data on medication-taking behaviors. With its user-friendly design, compatibility with various oral medications, and integration with MEMS AS software, the MEMS Button is a valuable addition to any clinical trial seeking to optimize patient outcomes and maintain research integrity.
What is a medication event monitoring system?
A medication event monitoring system (MEMS) is an electronic system used to monitor patient adherence to medication regimens. It typically employs medication adherence packaging and devices to record the date and time medication is dispensed or consumed. MEMS technology accurately tracks medication-taking behavior, enabling researchers to assess patient adherence patterns and make data-driven decisions to improve medication adherence. By providing a detailed record of when medication is taken, MEMS systems can also help identify barriers to adherence and facilitate targeted interventions to support patients adhering to their prescribed medication regimen.
Features of MEMS® Button Medication Event Monitoring System

FEATURES
Monitor Medication Events in Real-Time.
Real-time insights into adherence behaviors are critical for promptly identifying and addressing adherence issues. With the MEMS Button and its integration with AARDEX Group's Medication Adherence Software (MEMS AS), clinical researchers can access valuable real-time data on participant medication-taking behaviors. The user-friendly software includes pre-built dashboards and ranked lists that enable researchers to quickly identify participants struggling with adherence. With these insights at their fingertips, researchers can take appropriate measures to ensure that participants stay on track with their medication regimens, including providing additional support and coaching to those needing it most. With the ability to intervene early and proactively address adherence issues, researchers can optimize participant outcomes.
- Fully Scalable
- ISO27001 Certified Datacenter
- HIPAA & GDPR Compliant
- IRT, EDC & DCT Integration

FEATURES
Designed to the Highest Standard.
The MEMS Button has earned the prestigious CE mark, indicating that it adheres to the stringent health and safety standards established by the European Union. This certification confirms that the MEMS Button has undergone rigorous testing and meets the highest standards for safety and performance as a medical device. This achievement assures researchers of the accuracy and reliability of the MEMS Button when monitoring medication adherence in clinical trials. The CE mark also demonstrates our unwavering commitment to the safety and effectiveness of our products, providing reassurance to participants and sponsors alike. By prioritizing safety and efficacy, we strive to help researchers produce reliable and accurate study results, advancing the field of clinical research.
- Wireless Data Transfer
- 18 Month Battery Life
- Compatible with Most Medication Packages

FEATURES
Elevate Patient Adherence with MEMS® Mobile app.
Ensuring medication adherence is crucial for clinical trials to produce reliable and accurate results. We understand the importance of providing user-friendly solutions requiring minimal participant effort. Our solutions, including MEMS® Mobile app, allow participants to easily manage their medication schedules, helping them stay on top of their dosing regimen. With our patient-centric approach to medication adherence, we strive to empower participants to achieve better outcomes and ultimately improve their quality of life. We believe that by eliminating barriers to adherence, we can optimize participant outcomes and maintain research integrity in clinical trials.
- User-Friendly App
- IoS & Android Compatible
- Available in 25 Languages
- 20K+ Users in 30 Countries

OUR CLIENTS
The Go-To Solution for Pharma Companies
Some of the world's leading pharmaceutical companies have embraced our medication adherence solutions. From global giants to niche players, these organizations have recognized the value of our innovative solutions for enhancing medication adherence, reducing costs, and improving patient outcomes. It's an honor to partner with these remarkable brands, and we're proud to contribute to their efforts in advancing healthcare.

Medication Adherence Software →
Learn about our industry-leading adherence software for trials.

Medication Adherence Packaging →
Discover our range of medication adherence packaging.

Discover our range of medication adherence devices.
Got Questions?
Connect with an adherence expert.
Frequently Asked Questions
Medication adherence is a vital yet often overlooked aspect of successful research. That's why we've gone the extra mile to gather and organize the most frequently asked questions about this critical topic. Our goal is to empower researchers and patients alike with the knowledge they need to ensure medication adherence is never a hurdle to progress. So, without further ado, here are the answers you've been looking for!
In the context of medication adherence monitoring, a medication event refers to the act of taking medication according to the prescribed regimen. For instance, when a patient takes a pill, injects a medication, or applies a transdermal patch, it is considered a medication event. Monitoring medication events can provide valuable information about a patient’s adherence to their treatment regimen, such as the timing, frequency, and dosage of medication taken. This information can be used to identify patterns of non-adherence and to develop strategies for improving medication adherence in the future.
Medication event monitoring is important for several reasons:
Accurate tracking of medication adherence: Medication event monitoring systems accurately record when medication is taken. This can help healthcare providers, and researchers assess patient adherence patterns and identify potential barriers to adherence.
Improved patient outcomes: By monitoring medication events, healthcare providers can identify patients who are not adhering to their medication regimen and provide targeted interventions to support adherence. This can help improve patient outcomes and prevent complications related to non-adherence.
Better data for clinical trials: Medication event monitoring can improve the quality and reliability of data collected during clinical trials by providing more accurate and detailed information about medication adherence. This can help researchers make more informed decisions about the safety and efficacy of new treatments.
More efficient care: By identifying non-adherence early, researchers can take steps to address the issue and avoid unnecessary treatment modifications, hospitalizations, or additional medical expenses.
Although the terms “compliance” and “adherence” are sometimes used interchangeably, there is a subtle difference in their meanings:
Patient compliance generally refers to a patient’s willingness to follow a healthcare provider’s recommendations or instructions, including medication regimens, lifestyle modifications, or follow-up appointments. Compliance implies a certain degree of control or authority on the part of the healthcare provider, with the patient being expected to comply with the prescribed treatment plan.
On the other hand, patient adherence emphasizes the patient’s active participation in the treatment process, including their willingness and ability to follow the prescribed medication regimen, lifestyle changes, or self-care practices. Adherence implies a shared responsibility between the patient and the healthcare provider, with both parties working together to achieve optimal treatment outcomes.
In summary, while compliance and adherence refer to a patient’s ability to follow prescribed treatment plans, compliance emphasizes the provider’s authority and the patient’s willingness to comply. In contrast, adherence focuses on the patient’s active participation and shared responsibility in treatment.
WEBINAR WITH MERCK & BIOGEN
Mitigating the Risk of Poor Adherence in Trials
Watch this live recording with adherence experts from Merck & Biogen to learn about their approach to mitigating the risk of poor adherence in trials.


Collaborating for Safer, More Efficient Trials.
By combining technology and partnerships, we are revolutionizing how medication adherence is monitored in clinical trials. Our unique adherence ecosystem brings together leading medication adherence packaging and devices and DCT, IRT, and EDC vendors, CROs, and CMOs to drive innovation.
