According to the World Health Organization, up to half of the patients with chronic diseases fail to take their medications properly.1 Research shows many patients who are part of clinical trials do not maintain medication adherence either.2
Among many things, poor patient adherence is an ongoing threat to drug development. Typically, by month 12, 40% of participants have stopped taking treatment in line with protocol specifications, and a further 15% implement the study dosing regimen poorly.2
Historically, protocol deviations in relation to patient adherence have gone undetected by the non-digital measures of adherence, such as pill counts, blood sampling, and patients’ self-reporting, as inherently, they lack the precision to do so.
Implications of Poor Patient Adherence
Patient adherence, which involves patients taking their medications in accordance with how they have been prescribed by their healthcare provider, is a widespread challenge, a challenge that comes with wide-reaching implications for patients and health services worldwide. These include:
- Severe health complications.
- Premature deaths.
- Increased healthcare services demand.
- Wasted medications; and
- Lower quality of life.
The Cost of Poor Patient Adherence
It’s estimated that poor patient adherence contributes to almost 200,000 premature deaths across Europe each year. In the meantime, the annual cost of dealing with avoidable hospitalizations, emergency care, and outpatient treatment reportedly currently stands at around €125 billion in Europe and $105 billion in the U.S.3
Patients, healthcare providers, and other medical professionals can be negatively impacted by poor patient adherence. For patients, their health can be impacted by not taking the medication that has been prescribed to treat their health condition. While healthcare providers working to establish the effectiveness of the medication being used, their efforts can become compromised when patient adherence dwindles.
In drug development, key risks associated with poor adherence to study medications include underestimated drug efficacy to the point of trial failure, delayed approval of the investigational product, compromised patient safety, unduly large sample sizes, and flawed regulators’ benefit-risk assessment at the time of the drug being registered.2
Causes of Poor Patient Adherence
Patient adherence is a global medical problem mainly due to the fact it is prevalent in so many patients worldwide. And it’s a problem that can rapidly become increasingly complex due to the many different variables—for instance, the different patient views and attributes, illness characteristics, and service issues—that are all at play at any given time.
Some of the most common culprits that prevent patient adherence to medication from taking place include:
- Poor communication between the healthcare providers and patients.
- The patient having limited knowledge of the drug and how to use it.
- Fear of suffering adverse effects or side effects.
- The patient not being in agreement that they need the medication.
- Complicated dosing regimens.
- No perceived benefit from the medication.
- Forgetting to take the medication.
It’s widely reported that lack of clear instruction from healthcare professionals to patients, and the patient’s ability to understand the directions that they are asked to follow, is one of the most common stumbling blocks to optimal patient adherence.
Studies have found that between 40 and 60% of patients could not correctly report what their physicians expected of them 10 to 80 minutes after they had been provided with the information. Furthermore, more than 60% of patients interviewed immediately after visiting their doctor had misunderstood the instructions they had been given in relation to taking their prescribed medication.
Patient age has also been found to influence levels of patient adherence. For instance, medical intake among children is often impacted by the instructions and guidance that are provided by an adult, i.e. their parent(s) or care provider. While industry research has reported that patient among elderly patients stands at around less than 45%.
Measuring Patient Adherence
Given the widespread financial and practical implications of poor patient adherence, several measures have been developed and used over the years to help monitor and alleviate the issue.
These measures include:
- Direct methods—such as direct observed therapy, measuring the level of medication and blood or urine testing; and
- Indirect methods—such as patient questionnaires and self-reporting, pill counts, rates of prescription refills, assessments of patients’ clinical responses, measuring physiologic markers, and digital monitors (smart packaging).
The Efficacy of Traditional Approaches for Measuring Patient Adherence
Pill counts
In terms of the effectiveness of some of these methods, pill counts, which involve counting the number of tablets that are left in patients’ bottles or vials is simple. However, it can quickly become compromised if patients switch between medicine bottles or remove some tablets before their hospital visits to give the illusion of complying with their medication regimen. Furthermore, it’s not possible to monitor medication dose timing, which is a crucial factor that can help determine clinical outcomes.
Prescription Refill Rate
Prescription refilling rate is an imprecise measure of patient adherence and is only available if it is carried out by a closed pharmacy system. This limitation is because refills need to be counted several times. Furthermore, it does not provide the granularity of daily dosing which is crucial in understanding the impact of missed doses.
The Assessment of patients’ and clinical responses and measuring physiologic markers are too sparse to capture behavior and thus of limited use in practice and research.
Digital Patient Adherence Monitoring – an Evidence-Backed Approach
Of all of the measures to have been implemented so far, digital patient adherence monitoring is widely recognized as enabling the most objective and precise form of adherence monitoring to be achieved.
Generally speaking, the digital compilation of dosing history data is enabled by smart packaging and is a highly effective way to monitor, identify, manage and document the risks associated with poor patient adherence to medications in clinical trials.
This form of monitoring patient adherence typically involves an electronic microcircuit being placed within the medication packaging, which automatically generates a timestamp each time the patient takes their medication.4 The timestamps are then stored within the packaging’s ‘memory’ and then wirelessly transferred to a central, cloud-based software system where the patient adherence data can be analyzed.
The captured data is used to provide a proxy for drug intakes. In turn, this enables the patients’ actual medication adherence to be mapped and graphs and charts and other forms of tangible analysis to be produced that illustrate a patient’s medication intake over a given period. An example of this is shown in Table 1, where a dot displays a dose taken and a vertical line represents a day of missed dosing.
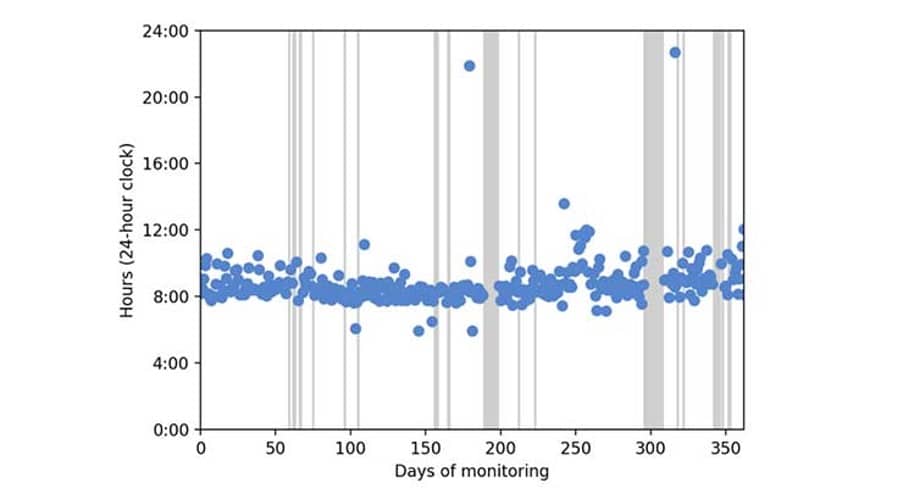
Table 1. Example of data produced by monitoring of a subject’s medication intake digitally for 363 days.
Detecting changes in Patient Adherence
Aside from providing an overview of patients’ medication adherence, state-of-the-art pioneering digital monitoring systems, which incorporate online algorithms, provide the ability to drill down that bit further and truly understand patients’ medication-taking habits, as well as to detect any potential issues.
Segmenting and understanding different Patient Adherence behaviors
Assessing behavior changes provides homogeneous behavior periods that can then be characterized using a representative adherence measure. This way of working provides an appropriate summary of a patient’s adherence, which hasn’t been oversimplified by using a single adherence metric that’s computed on the whole monitoring period.
As a consequence, using single metrics to understand patients’ medication intake can often confuse the analysis and interpretation of the results. This is because:
- The patients’ medication taking-behavior can change—it’s possible for it to get better or worse and continuously fluctuate over the period of time it is being monitored; and
- Simple percentage statistics don’t distinguish between the different non-adherent behaviors that can range from perfect adherence to drug holidays and regular drug omissions. All of these behaviors have the potential for the drug that’s been administered to lead to very different effects, depending on how it is, or isn’t, being used.
Typical behavior changes include drug holidays, weekend drop-offs, different intake times, etc. It’s also possible to highlight several successive behavior changes, as well as patients whose behavior remained fairly consistent.
Detecting Patient Adherence Issues Early On
Monitoring patient adherence also provides an invaluable opportunity to identify any key behavior changes the moment they happen. This is essential in detecting any issues that may require healthcare or professional medical support early on.
For example, you could take the data from patients whose medication adherence for a daily anti-cancer drug is being monitored between six and 421 days. The algorithm is then able to detect the behavior changes during the monitoring period triggering a healthcare provider support focus discussion with the patient.
For instance, because the algorithm is online, it can be applied during ongoing patient monitoring and send an alert to their subject’s healthcare provider or study coordinator the moment a behavior change has been detected.
This trigger then enables the healthcare provider to reach out to the patient to understand what has caused the behavior change (e.g. if they’ve been experiencing side effects). If the new behavior has a worse adherence than the previous one, then the healthcare provider can advise the patient to switch back to their previous medication intake habit, as well as provide them with the guidance and instructions that will enable them to do so effectively.
Managing Patient Adherence in Clinical Trials
Medication adherence management methods can single-handedly improve clinical results and conclusions. However, digital monitoring technology further enables:
- Drug developers or clinical study supervisors to achieve true optimization for their drug development, using proven and effective monitoring of patient non-compliance within their clinical trials; and
- Brand managers or innovation packaging managers to help their organization stand out and differentiate themselves against their competitors by taking the additional steps to improve the safety and efficacy of their therapies
The ability to measure patient signals and automatically or promptly administer, medication provides a closed feedback loop that has the potential to optimize effectiveness for individual patients, ensure the robust continuation of medication and optimize patients’ outcomes.
Non-digital methods, such as pill counts, blood sampling, and patients’ self-report, may be widely used, but they are not accurate, tend to overestimate medication intake, and can bias the results of adherence research.
Overall, digital monitoring is at the cornerstone of new technology adoption to replace unreliable methods to assess patient adherence. This is especially true given that today’s industry-leading platforms can:
- Optimize drug development using adherence-informed clinical trials;
- Ensure high fidelity to the dosing regimen specified in the clinical trial protocols, as suggested; by the U.S. FDA and EMA
- Identify structural deficiencies during clinical trials;
- Gather key patient adherence behavioral data for successful marketing strategy;
- Improve medication adherence, data quality, and integrity; and
- Support proactive evidence-based risk mitigation strategies.
Smart Packaging = 97% accuracy
Advanced digital patient adherence monitoring provides complete oversight of the adherence metrics and risk indicators that matter the most. According to studies, smart packaging/device monitoring is 97% accurate, ahead of drug levels and markers (70%), pill counts (60%), healthcare professional ratings (50%) and patient self-reporting and electronic patient diaries (27%).
Smart packaging is frictionless and thus more effective compared to using just app-based methods. App-based solutions may struggle to gain compliance from non-adherent types compared to if you were using passive measurements from smart packaging, which will capture both adherent and non-adherent types of patients.
Digital Times Call for Digital Patient Adherence Monitoring
Particularly state-of-the-art measures that have been designed to deliver clinical trial success by using reliable, up-to-the-minute, evidence-based tactics. These measures have been honed and perfected over several product generations.
Such digital solutions not only provide more reliable data, but also more detailed data about actual patient adherence, such as dose frequency, dose interval, and medication dose timing—details that traditional methods do not capture. Digital monitoring shows that poor adherence, especially dosage omission or changing intervals, is more prevalent than previously recognized.
These smarter monitoring systems enable sponsors to improve drug efficacy by managing patient adherence to the study medications. With digital medication adherence monitoring, pharmaceutical companies can safeguard their clinical trials and build a stronger argument about the efficacy of their medication from the development stage to commercialization.
References
- https://www.who.int/chp/knowledge/publications/adherence_report/en/
- Adherence to medications: insights arising from studies on the unreliable link between prescribed and actual drug dosing histories. Blaschke TF, Osterberg L, Vrijens B, Urquhart J. Annu Rev Pharmacol Toxicol. 2012;52:275-301
- https://www.oecd-ilibrary.org/social-issues-migration-health/investing-in-medication-adherence-improves-health-outcomes-and-health-system-efficiency_8178962c-en
- https://www.oecd-ilibrary.org/social-issues-migration-health/investing-in-medication-adherence-improves-health-outcomes-and-health-system-efficiency_8178962c-en
About the Author
Bernard holds a PhD from the Department of Applied Mathematics and Informatics from Ghent University, Belgium. As Scientific Lead at AARDEX ® Group, Bernard oversees the research and development of sophisticated analytical methods for monitoring participant medication-taking behaviours.
Along with providing guidance on adherence to Pharmaceutical, BioPharma, and Academic organizations, Bernard is a founding member of the International Society for Patient Adherence (ESPACOMP) and is an active member of several EU and US-funded consortiums that focus on adherence. Having co-authored several book chapters, over 100 peer-reviewed scientific papers, Bernard is considered an expert in his field.



