Abstract
Objective
To assess methotrexate (MTX) adherence using the Medication Event Monitoring System (MEMS) and characterize associations with adherence in patients with rheumatoid arthritis (RA).
Methods
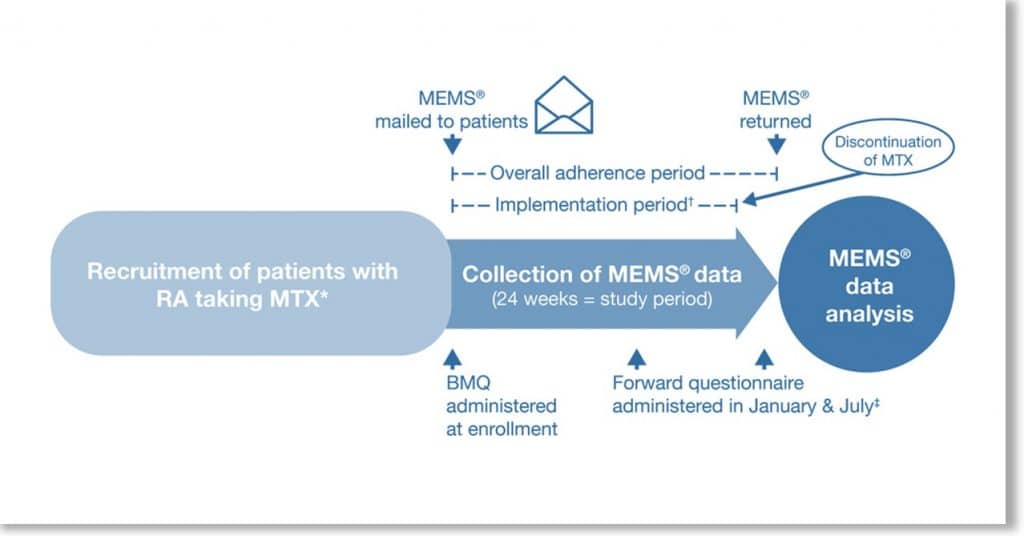
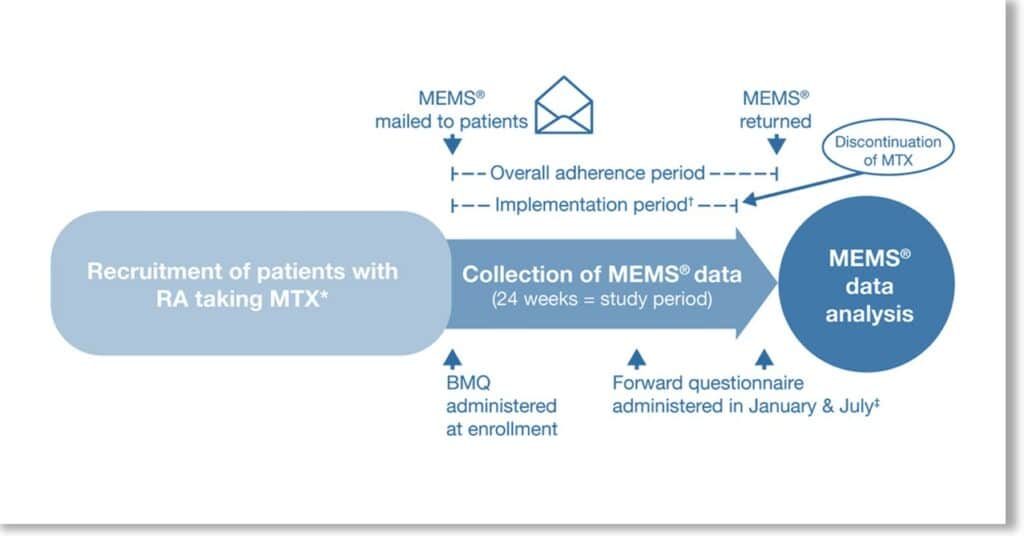
Eligible patients participated in Forward, the National Databank for Rheumatic Diseases, and recently (12 months or sooner) initiated oral MTX. MEMS was used to compile MTX weekly dosing over 24 weeks. The Beliefs about Medicines Questionnaire (BMQ) was completed, and baseline demographics and disease characteristics obtained. MTX adherence (percentage of weeks dose taken correctly), implementation (percentage of weeks dose taken correctly from initiation until last dose), and persistence (duration from initiation to last dose) were calculated. Analyses measured associations between patient characteristics and adherence, modeled using logistic generalized estimating equations and censored Poisson regression, and persistence modeled using Cox regression.
Results
Overall, 60 of 119 eligible patients were included in the analysis. MTX adherence, implementation, and persistence were 75%, 80%, and 83%, respectively, at 24 weeks. Demographics and disease characteristics were generally similar between patients with 1 week or less and 2 weeks or more of missed MTX. Unemployment, less disability, higher Patient Global scores, and no prior disease‐modifying antirheumatic drug (DMARD) use were associated with correct dosing. No significant differences in adherence were observed between patients receiving concomitant MTX versus MTX monotherapy, and biologic DMARD‐experienced versus biologic DMARD‐naïve patients. Higher scores in BMQ Specific Necessity (indicating a greater belief in the necessity of the medication) was associated with a decreased likelihood of dosing at an interval shorter than prescribed (odds ratio 0.89).
Conclusion Even in a participatory group over a short period, MTX adherence was suboptimal and associated with certain demographics, medication experience, and beliefs about medicines. This suggests a need for screening and alternative treatment opportunities in nonadherent MTX patients with RA.
View the complete publication on : https://onlinelibrary.wiley.com/doi/full/10.1002/acr2.11079